| Reprinted from Excerpta Medica International Congress Series No.87 PROCEEDINGS OF THE XXIlIrd INTERNATIONAL CONGRESS OF PHYSIOLOGICAL SCIENCES Tokyo, September 1965 |
| BEHAVIOURAL AND EEG EFFECTS OF PARADOXICAL SLEEP DEPRIVATION IN THE CAT |
| M. JOUVET , School of Medicine, Lyon, France |
Since the work of Pieron (1913) 18, the effect of sleep deprivation has been largely studied both in man and animaIs. But the problem of sleep has become complicated recently by the demonstration that sleep could not be considered as a unique phenomenon (opposed to wakefulness), but on the contrary was composed of two different neural states, being most probably the expression of different mechanisms: sleep with synchronized EEG activity = slow sleep (S.S.) and sleep with desynchronized EEG activity (activated sleep: Dement, 1958 2; paradoxical sleep (P.S.); Jouvet, Michel and Courjon, 1959 14).
This report will show some behavioural and EEG effects of P.S. deprivation in the cat. After a summary of recent findings concerning P.S., the effects of temporary instrumental deprivation of P.S. both in normal and chronic pontile cats wil1 be shown and final1y we shal1 describe the striking behavioural disturbances which periodical1y occur in cats in which P.S. is definitively suppressed by limited pontile lesions.
I. PARADOXICAL SLEEP
Paradoxical sleep, in normal cats, always follows a period of slow wave sleep. Its mean duration is 6 minutes. It is electrophysiologically characterized mainly by fast cortical EEG activity, almost similar to arousal, by a regular theta rhythm in both dorsal and ventral hippocampus, and by monophasic spikes occurring in the reticular formation of the pons (Jouvet et al., 1959 14), in the lateral geniculate nucleus (Hendley, 1963 5), and in the occipital cortex (Mouret, Jeannerod and Jouvet, 1963 17). This phasic activity is the first sign of P.S. and may precede cortical activation by a few minutes. Behaviourally, the total atony of the neck muscles (Jouvet et al., 1959 14) and rapid eye movements (Dement, 1958 2) which patterns are different from waking (Jeannerod and Mouret. 1963 7) and clonic jerks are the most salient features of this state of sleep which represents 20 to 25% of total sleep (and therefore 15% of the day).
P.S. is considered to be the deepest state of sleep since the threshold of arousal by stimulation of the midbrain reticular formation is much enhanced in comparison with S.S. (Jouvet et al., 1959 14). It has been shown that P.S. may be triggered by stimulation of the brainstem at high frequency (Jouvet, 1962 10), and that pontine reticular structures are necessary and sufficient for its occurrence, since P.S. stil1 occurs periodical1y in chronic pontile cats (but not in retropontine animaIs) whereas limited destruction of the pontine reticular formation electively suppresses P.S. without suppressing waking or S.S. (Jouvet, 1962 10). Two main problems are stil1 not solved concerning this state of sleep: the first one concerns the specificity of P.S.: for some authors, it is believed that P.S. and S.S. are the expression of the same hypnogenic mechanism and that P.S. is only due to the exaggeration of the mechanism responsible for S.S. ln this meaning, P.S. would appear as a deep sleep opposed to S.S. considered as a light sleep (Hernàndez-Peon, 1965 6). On the contrary, the hypothesis that P.S. is a state qualitatively different from S.S. has been put forward on the basis of electrophysiological (Jouvet, 1962 10), ontogenetic (Valatx, Jouvet and Jouvet, 1964 21) and phylogenetic (Klein, 1963 15) studies. The second problem concerns the mechanisms and the function of P.S. which are a!most unknown. It has been suggested that P.S. could depend upon a neurohumoral mechanism located in the pontine R.F ., but the neurohumoral transmit ter is still unknown. Some behavioural aspects of P.S. seem to favour a cholinergic mechanism: fissurated myosis, fall of blood pressure (Rossi, 1963 20), inhibitory effect of atropine (Jouvet, 1962 10), relaxation of nictitating membranes, whereas the inhibitory action of reserpine which DOPA may counteract is in favour of catecholaminergic mechanisms (Matsumoto and Jouvet, 1964 16). On the other hand, the fact that y-butyrolactone (y HL) and y-hydroxybutyrate of sodium may specifically induce P.S. in normal, decorticate or pontile cats (Jouvet, Cier, Mounier and Valatx, 1961 12) has not yet received any explanation since it is still not certain whether y HL is a normal constituent of the brain (Bessman and Fishbein, 1963 1; Giarman and Roth, 1964 4). Specific deprivations of P.S. have thus been carried out in order to bring more facts to contribute to the understanding of the mechanisms and function of P.S.
Il. INSTRUMENTAL DEPRIVATION OF PARADOXICAL SLEEP
(a) Normal cats
A technique of instrumental and selective deprivation of P.S. (Jouvet, Vimont, Delorme and Jouvet, 1964 9) was employed in order to dissociate the two states composing behavioural sleep in normal chronic cats with implanted electrodes. The animal is placed on a small support floating on water. It can stand or crouch but the small surface of the support prevents it from Iying down completely and relaxing its muscular tonus without falling into the water. The EEG and EMG activity is recorded continuously or integrated by means of a special device (Oneirograph: Jouvet, 1962 11). The animal's behaviour is further recorded with the aid of photographs. Six cats were subjected to successive periods of deprivation of 1, 2, 3, 4, 5, 6, 9, 10, 15, 22 and 26 days. At the end of depfivation, the cats could recover normal sleep in sound-proof cages and were registered continuously until the control level of sleep-waking rhythm reappears.
Results of deprivation
1. During the deprivation process
At the beginning of deprivation, behavioural and EEG arousal was slightly increased (40-60% ) as a result of agitation, but phases of S.S. reappeared within one day. Spindles and slow waves appeared in the cortex and in subcortical structures, and some monophasic spikes in the pontine reticular formation and in the lateral geniculate body, while the neck of the animal flexed. These phases of S.S. were always followed by a sud den arousal caused by the loss of balance as the neck bent more and more. Hehavioural or EEG P.S. is thus impossible, and whilst deprivation of P.S. is absolute, deprivation of S.S. is only minimal (10-20% depending on the animal).
After longer deprivation (more than one week), changes in sleep-waking behaviour appear. S.S. is almost continuous but it never lasts for more than 2 or 3 minutes since it is interrupted by the fan of the animal's head or body into the water, and thus by a short arousal. The circadian duration of S.S. decreases by 20-30% due to the more frequent arousal. Tachycardia (60-80% increase of control heart rate) appears at the beginning of deprivation. It lasts continuously and the heart rate does not decrease during S.S. When the animaIs are observed outside the tank, they look more and more sleepy, although S.S. deprivation is only relative. They are inert, exhibit a pronounced photophobia, eat and drink more. They have a muscular weakness and hypotonia and they cannot usually jump upon a chair. Even during the longest deprivations, we have not seen hallucinatory-like behaviour such as described in cats with pontine lesions (see below).
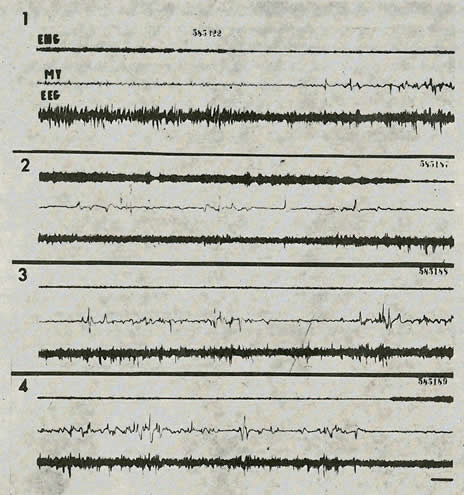
Fig. 1. Narcolepsy during recuperative sleep. Recuperative sleep after 26 days of P.S. deprivation.
- (1) Normal pattern - sequence of S.S. and P.S. with rapid eye movements (M.Y.) and occipital spikes (BBG).
- (2, 3, 4) Narcoleptic pattern - the tracings are continuous; direct transition from waking (2) to P.S. (eye movements and occipital spikes) (3) and return to waking (4). There is no intermediary S.S.
Each tracing represents 2 minutes of recording. Scale: 6 sec. - 50 microvolts.
2. During recuperation
Recuperative phases were identical in aIl the animaIs. On leaving the tank, after a short period of grooming, the animaIs fall into P.S.
After short deprivations, there was always a period of S.S. of short duration before the P.S., but after deprivations longer than one week, P.S. could appear immediately after waking without any S.S. (Figure 1). This fact recalls narcolepsy which EEG has been described in man by Rechtschaffen, Wolpert, Dement, Mitchell and Fisher (1963) 19.
During the 6 first hours of recuperative sleep, there was avery marked augmentation of P.S. (up to 60% of total sleep time). This ratio, reached after 3 days of deprivation, was not exceeded even after a deprivation lasting for 22 days. It seems thus to repre5ent a biologicallimit in the adult cat.
This high percentage of P.S. is due to a slight increase in its average duration (8 minutes against 6 minutes 20 seconds in controls) and especially to the shorter intervals between the phases of P.S. The animal never recovered in only one long P.S. episode and the process of P.S. recovery was always periodically interrupted by periods of s.s. During the first episodes of recuperative P.S., the phasic movements (paws, tail, whisker, eyes) were much increased and so intense that the animal occasionally presented a picture of epileptic seizures. There was also an increase of monophasic spikes in the pontine and lateral geniculate regions. At these times, the arousal threshold was very high and nociceptive stimuli were necessary to awaken the animal.
This explosive return of P.S. after deprivation th us suggests a phenomenon of rebound and elective recuperation. The duration of the increase of P.S. during recuperation is proportional to the duration of the deprivation period (Figure 2). lt is equal to hal! o! the duration o! the deprivation, i.e. after a deprivation of 22 days, it will take Il days for a cat to corne back to a controllevel of 20% of P.S. (compared with total sleep). lt is interesting to note that the return to a normal heart rate follows the same rule (Figure 2).
Thus, it appears that P.S. deprivation may bring long-Iasting changes both in sleep mechanisms and in the vegetative system. Death occurred in one animal after 26 days of P.S. deprivation, on the third day of recovery. The recuperative sleep was very ab normal and there were many episodes of cataplexy with suddèn postural collapse, atonia, without rapid eye movements. The heart rate did not decrease during the recovery and ventricular fibrillation occurred. There were lesions in the suprarenal glands.
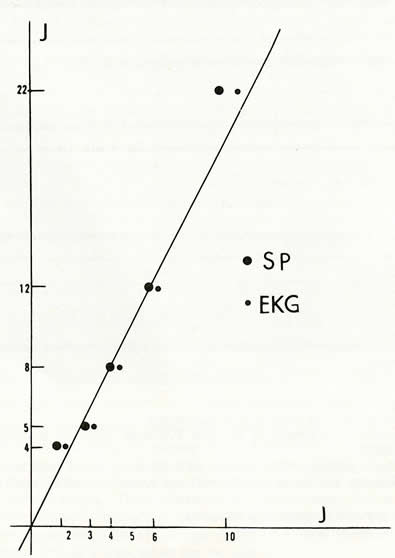
Fig. 2. Duration of the period of recovery for P.S.
and heart rate after deprivation. in ordinates: duration of P.S. deprivation
(in days). in abscissae: duration of the recovery period (during which
there is an increase of P.S. and heart rate) (in days).
big dots: duration for P.S.
small dots: duration for heart rate.
Effect o! pentobarbital
The process of P.S. recuperation after selective deprivation is the expression of a very active mechanism since it is not suppressed by pentobarbital, even with an anaesthetic dose (Jouvet and Delorme, 19648). As a mat ter of fact, injection of 30-35 mg./kg. of pentobarbital in a cat, at the beginning of recuperative sleep (after more than 3 days of P.S. deprivation) does not suppress the periodical appearance of bursts of monophasic pontine or geniculate spikes, although there is no EEG activation, at the beginning. Paradoxically, it was found that the cortical activation pattern of P.S. appears, after deprivation, earlier under pentobarbital narcosis than in normal conditions. The "need" for P.S. appears thus to counteract the effect of narcosis.
(b) Chronic pontile cats
In 20 animaIs, after total section of the brainstem in front of the tentorium (level Az of the Horsley-Clarke coordinates), the cerebral hemispheres, the limbic system and thalamus are removed by aspiration. A hypothalamic island is left in situ. The brainstem transection between the pons and the remaining hypothalamic island is then completed by aspiration and an acrylic strip 1-2 mm thick and 15 mm wide fixed dorsally to the tentorium cerebelli is wedged between the brainstem section surface and the remaining hypothalamic island in such a manner as to prevent any possible neurocrine connections between the hypothalamus and the brainstem (Figure 3). Such chronic preparations in which pontine electrical activity and EMG activity are continuously recorded, were kept, for as long as 3 months, in incubators at normal rectal temperature. They exhibited only two different states: waking and P.S. (Jouvet, Jouvet and Valatx, 1963 13). P.S. occurs regularly like a biological clock. Its duration is the same (6 minutes) as in normal cats whereas its circadian duration is smaller (10% of the day versus 15% in normal cats).
P.S. is characterized by the occurrence of monophasic spikes in the pontine reticular formation which pattern is the same as that recorded at the same level in the normal animal, by the sudden disappearance of muscular tone, by eye movements (lateral, i.e. dependent on the VIth nerve), and by vegetative variations: irregularity of respiration, acceleration of heart rhythm. P.S. occurs immediately after "waking" since there is no behavioural or EEG subcortical period of slow sleep in these preparations.
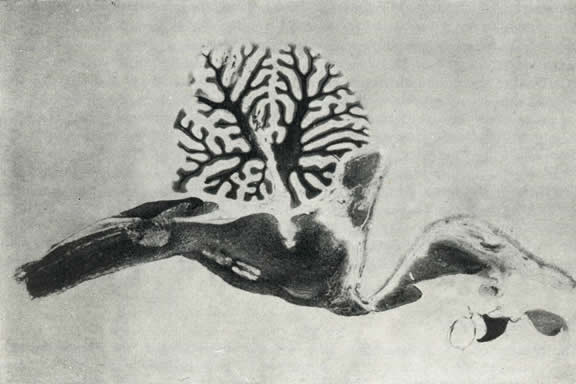
Fig. 3. Medio-sagittal section of the brain of a pontile
animal with hypothalamic island.
Anteriorly, the trace of the brainstem section can be seen where the strip
of acrylic resin was placed, and posteriorly, the trace of an electrode
in the caudal part of the nucleus reticularis pontis caudalis.
Animal sacrificed on the 66th day. Luxol staining.
Deprivation of P.S.
Our technique was as follows: at the beginning of each phase of P.S., the fall in nuchal EMG activity, by the intermediary of an integrator, switches on a stiroulator which applies electric shocks to the animal's leg. The strength of these shocks is regulated to arouse the animal in which case the reappearance of EMG activity automatically cuts out the stimulation. Deprivation was maintained for a maximum of 8-9 hours. It appeared that when P.S. is suppressed by a shock, it tends to recur at ever shorter intervals. At first these intervals are siroiiar to those between periods of P.S. in controls (i.e. 40-45 minutes), but after some hours they become so short that 40-60 seconds after being wakened by the shock, the animal falls into a new phase of P.S. (Fig 4). Thus the total number of incipient phases of P.S. was as rouch as 155 in sessions that lasted for 9 hours and P.S. can hardly be suppressed for longer periods than this in view of the very great number of shocks required. When the stimuli were stopped, P.S. returned immediately: its average duration was then only slightly more than that controls (7 as against 6 minutes), and its periodicity reroained at a higher level for 2-3 hours and then returned to normal.
A result such as this exeroplifies the necessary occurrence of P.S. in pontile animaIs and demonstrates that there is also a "need for P.S." in such preparations. This strongly suggests that P.S. represents an active process situated in the lower brainstero the function of which is still unknown.
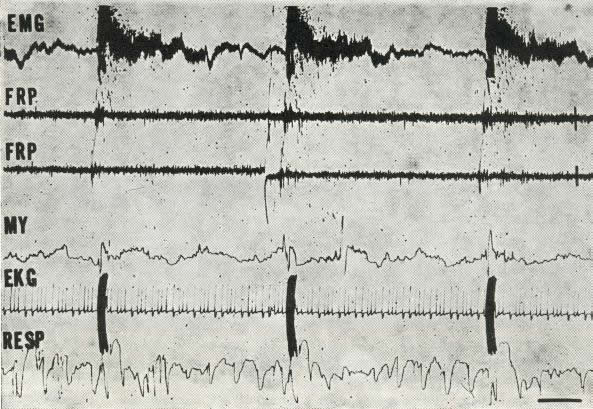
Fig. 4. Automatic deprivation in a pontile cat. Slow
speed recording.
Disappearance of EMG activity in the neck on incipient P.S. produces an
electric shock (artefact) which wakes the animal for some 10 seconds until
P.S. inevitably sets in once again (5th hour of deprivation).
F .R.P . - Activity of the pontine reticular formation.
Note the appearance-of eye movements (M.Y.) as EMG activity falls.
Scale: 3 sec. - 50 microvolts.
III. DEFINITIVE SUPPRESSION OF P ARADOXICAL SLEEP WITH CIRCUMSCRIBED LESION OF THE PONTINE RETICULAR FORMATION
Whereas coagulations of the medial pontine reticular formation (nuclei of the raphe) or of the medial part of nuclei reticularis pontis oralis or pontis causalis do not induce any significant change in both waking, 8.8. and p .8., limited lesions of the dorsolateral part of the pontine reticular formation totally suppress both total atonia and fast activity during behavioural sleep. However, there is still alternation of EEG and behavioural waking and 8.8. (Jouvet, 1962 10.11). There is also a marked increase of lateral geniculate spikes which occur during 8.8. almost continuously. ln such cats, the injection of r-butyrolactone or sodium r-hydroxybutyrate is unable to induce p .8. (Jouvet et al., 1961 12). In these cats very striking and dramatic changes of behaviour occur periodically (and will be shown in an 8 mm movie). The most frequent pattern resembles hallucination; it appears one or two weeks after the lesion. 8.8. is interrupted by some jerks of the neck muscles and the cat suddenly exhibits anger, fear or rage or it may display a sexual behaviour (in the female cat). It stands on its legs and violently attacks imaiïnary enemies with its legs, or it may watch some unknown object for 1 or 2 minutes. Usually, during such a behaviour, the nictitating membranes are relaxed and may cover the pupils which are myotic. Thus, there is a dissociation between the behaviour which resembles rage and the ocular syndrome which resembles sleep. At this time there is a fast low-voltage cortical EEG activity with bursts of spikes in the lateral geniculate while the EMG activity of the neck muscles is enhanced (Figure 5). It should also be pointed out that a permanent tachycardia appears in these animaIs. Such "hallucinatorylike behaviour" periodically occurs during S.S. It is not suppressed either by light or darkness. This behaviour has been displayed in every animal (8 cats) in which P.S. was suppressed by pontine lesion. ln the case of recovery with subtotal lesion of dorsolateral pontine reticular formation, this behaviour disappeared when total atonia of the neck muscles and fast activity during sleep reappeared.
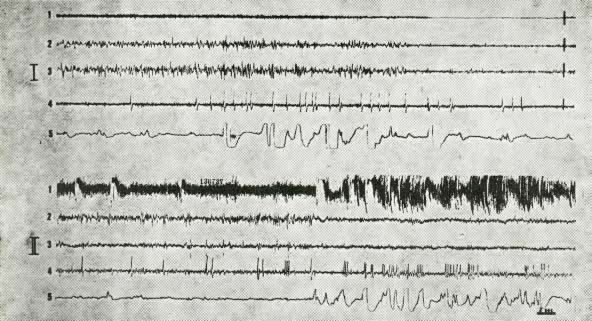
Fig. 5. Polygraphic aspects of hallucinatory-like behaviour.
I. Onset of P.S. before the coagulation of the pons.
Decrease and disappearance of EMG activity of the neck (1).
Fast low voltage cortical activity after a period of S.S. (sensorirnotor and occipital cortices - 2, 3 -).
High voltage monophasic spikes at the lateral geniculate nucleus (4).
Rapid eye movements (5).
II. Three weeks after lesion of the dorsolateral pontine reticular formation.
S.S. is suddenly followed by a "hallucinatory-like behaviour" with enhanced EMG activity, fast cortical activity, bursts of lateral geniculate spikes and rapid eye movements.
Slow speed recording.
Scale: 3 sec. - 50 microvolts.
DISCUSSION
All these results will be briefly discussed here. They suggest that P.S. is due to an active mechanism (which is resistant to pentobarbital anaesthesia) and which necessitates the integrity of the dorsolateral part of the pontine reticular formation. That there is a "need for P.S." is weIl illustrated by the fact that its suppression leads to an almost insuperable recurrence of P.S. (which is in agreement with Dement's experiment on dream deprivation in human) (1960) 3. It must be pointed out that, although S.S. is almost normal in duration in cats submitted to instrumental deprivation of P.S., a need for sleep increase8 during P.S. deprivation. One would have to assume that S.S. by itself is not sufficient to take on the overall "recovery function of sleep" or that P.S. might assume another function which cannot be accomplished by S.S.
The long-Iasting changes induced by long duration deprivations of P.S. suggest that some cerebral biochemical disturbances may occur which express themselves through a permanent tachycardia (of which central origin is likely).
P.S. being dependent upon some neurohumoral process would also explain the finding that recovery processes, after P.S. deprivation, are effected by a periodical mechanism (and not by a unique longer P.S. phase). This recovery sleep with its high ratio of P.S. appears to be the expression of some "eliminating" process requiring a given level of enzymes, which would have to be synthetized during the intervals between successive P.S. The fact that the return to a normal heart rhythm closely follows the recovery of a normal level of P.S. is in agreement with the fact that P.S. is dependent upon a certain amount of Dopamine or Noradrenaline in the brain (as shown by experiments with reserpine and DOPA) (Matsumoto and Jouvet, 1964 16). It should also be assumed that, when the pontine mechanisms (controlling total atonia and fast cortical activity during P.S.) are destroyed by a limited pontine coagulation, the increase of geniculo-occipital monophasic spikes might be the electrophysiological manifestation of some precursory mechanism of P.S. Such an increase of activity in the visual system would explain the hallucinatory behaviour displayed by cats with pontile lesions: these animais can no longer inhibit their muscular activity. ln such cases, this "hallucinatory-like behaviour" would appear to express some telencephalic "dreaming" while the rhombencephalic inhibitory mechanism, which normally acts upon the motor outflow during P.S., is suppressed.
Finally, the possible relationships between these states and some psychiatric disturbances in man must also be borne in mind.
REFERENCES
- 1. BESSMAN, S. P., and FISHBEIN, W. N.:
Gamma-hydroxybutyrate, a normal brain metabolite. Nature (Lond.), 1963, 200 (1207). - 2. DEMENT, W. C.:
The occurrence of low voltage, fast electroencephalogram patterns du ring behavioral sleep in the cat. Electroenceph. clin. Neurophysiol., 1958, 10 (291). - 3. DEMENT, W. C.:
The effect of dream deprivation. Science, 1960, 131 (1705). - 4. GIARMAN, N. J., and ROTH, R. H.:
Differential estimation of gamma-butyrolactone and gamma-hydroxybutyric acid in rat blood and brain. Science, 1964, 145 (583). - 5. HENDLEY, C. D.:
Sharp waves in lateral geniculate of cat with eye movements during low voltage sleep. Fed. Proc., 1963 (637). - 6. HERNÀNDEZ-PÉON, R.:
A cholinergic hypnogenic limbic forebrain-hindbrain circuit. In: Aspects anatomo-fonctionnels de la Physiologie du Sommeil, Jouvet, M., editor, C.N.R.S. Paris. 1965, in press. - 7. JEANNEROD, M., and MOURET, J .:
Etude comparative des mouvements oculaires au cours de la veille et du sommeil chez l'homme. I. Physiol. (Paris), 1963, 55 (268). - 8. JOUVET, D., and DELORME, J. F.:
Evolution de certains signes du sommeil paradoxal au cours de la narcose nembutalique chez le chat. C. R. Soc. Biol. (Paris), 1964, in press. - 9. JOUVET, D., VIMONT, P., DELORME, F., and JOUVET,
M.:
Etude de la privation sélective de la phase paradoxale de sommeil chez le chat. C. R. Soc. Biol. (Paris), 1964, 158 (756). - 10.
JOUVET, M.:
Recherches sur les structures nerveuses et les mécanismes responsables des différentes phases du sommeil physiologique. Arch. ital. Biol., 1962, 100 (125). - 11. JOUVET, M.:
Un appareil enregistreur automatique des phases rhombencéphaliques du sommeil chez le chat: l'onirographe. Rev. neurol., 1962, 107 (269). - 12. JOUVET, M., CIER, A., MOUNIER, D., and VALATX,
J. L.:
Effets du 4-butyrolactone et du 4-hydroxybutyrate de sodium sur l'EEG et le comportement du chat. C. R. Soc. Biol. (Paris), 1961, 155 (1313). - 13. JOUVET, M., JOUVET, D., and VALATX, J. L.:
Etude du sommeil chez le chat pontique. Sa suppression automatique. C. R. Soc. Riol. (Paris), 1963, 157 (845). - 14.
JOUVET, M., MICHEL, F., and COURJON, J.:
Sur un stade d'activité électrique cérébrale rapide au cours du sommeil physiologique. C. R. Soc. Riol. (Paris), 1959, 153 (1024). - 15. KLEIN, M.:
Etude Polygraphique et Phylogénétique des Différents États de Sommeil, Thèse de Médecine, Bosc, editor, Lyon, 1963. - 16. MATSUMOTO, J., and JOUVET, M.:
Action de réserpine, DOPA et 5-HTP sur les différents états de sommeil chez le chat. C. R. Soc. Riol. (Paris), 1964, in press. - 17. MOURET, J., JEANNEROD, M., and JOUVET, M.:
L'activité électrique du système visuel au cours de la phase paradoxale du sommeil chez le chat. J. Physiol. (Paris), 1963, 55 (305). - 18. PIERON, H.:
Le Problème Physiologique du Sommeil, Masson (Paris), 1913 (520 pages). - 19. RECHTSCHAFFEN, A., WOLPERT, E., DEMENT, W., MITCHELL,
S., and FISHER, C.:
Nocturnal sleep of narcoleptics. Electroenceph. clin. Neurophysiol., 1963, 15 (599). - 20. ROSSI, G. F.:
Sleep-inducing mechanisms in the brain stem. Electroenceph. clin. Neurophysiol., 1963, Suppl. 24 (113). - 21.
VALATX, J. L., JOUVET, D., and JOUVET, M.:
Evolution électroencephalographique des différents états de sommeil chez le chaton. Electroenceph. clin. Neurophysiol., 1964. 17 (218).