| Human insulin gene insertion in mice. Effects on the sleep-wake cycle? |
|
JEAN-LOUIS VALATX (1), PHILIPPE DOUHET (2) and DANIELLE BUCCHINI (3) J. Sleep Res. (1999) 8, Suppl. 1, 65-68 |
| Table of Contents |
| Printable version |
RESULTS
Experiment 1
Figure 1(A) shows that the amount of SWS over the 24-h period was significantly higher in transgenic (DELTA-168) mice (+ 15.60% + 0.7%, P < 0.001); this increase was mainly due to the increase in the amount of SWS during the night period. The amount of PS was not affected. After 10 h of sleep deprivation, the rebound was seen only during the first night and was lower in transgenic mice than in controls (SWS, + 71.1 + 16.6 min vs. + 136.7 + 5.6 min; PS, + 33.0 + 2.0 min vs. + 44.4 + 3.1 min), representing a sleep debt payback of 13.6% vs. 31.4% for SWS and 61.6% vs. 81.7% for PS (Fig. 2). During the second 24-h period, SWS and PS amounts were not different from controls.
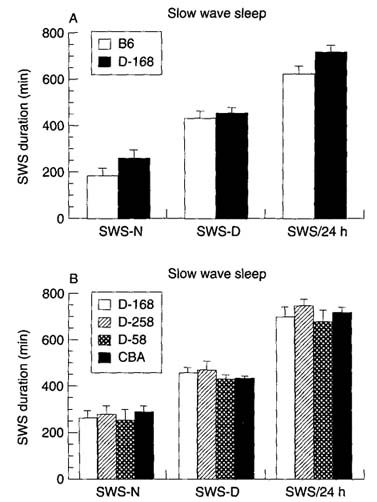
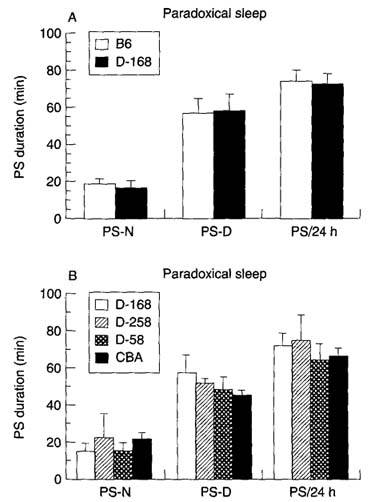
Sleep duration in transgenic mice. (A) SWS and PS during the night (SWS-N, PS-N) or day (SWS-D, PS-D) periods or over 24 h (SWS-24 h, PS-24 h) in DELTA- 168 (stippled bars, n = 9) and control B6 (white bars, n ~ 6) mice. (B) SWS and PS duration in mice expressing the human insulin transgene in the brain (DELTA-168) or in pancreatic islets (DELTA-258, n = 4), in mice not expressing the transgene (DELTA-58, n = 5), and in CBA mice (black bars, n = 10). Mean values + SD of the five baseline days.
Experiment 2
In DELTA-258 mice, the amount of SWS and PS during baselines (Fig. 1B) and recovery (Fig. 2) was not statistically different from that seen in DELTA-58 mice. In addition, neither strain showed a significant difference in these parameters compared with A168 mice, whereas both showed a significant difference when compared with control B6 mice. In both the DELTA-258 and the DELTA-58 strains, the rebound after sleep deprivation was not different statistically from that seen in DELTA-168 mice, but there was considerable variation between the individual mice in each strain (Fig. 2).
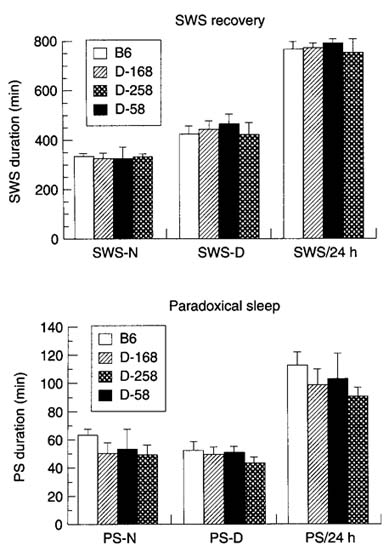
Recovery after a 10-h sleep deprivation period in control B6 mice (white bars) and in three transgenic mice with differing expression of the human insulin gene (see legend to Fig 1 B). The same mice as in Fig 1 were submitted to sleep deprivation. Mean values + SD.