| The Discovery of Cholera-Toxin as a Powerful Neuroanatomical Tool |
| by Pierre-Hervé Luppi , Ph.D. |
For thirty years, the advances made in our lab in neural mechanisms responsible for the generation of the sleep-waking cycle have been obtained by a multidisciplinary approach and the development of new methods of investigation. Indeed, physiological, neuroanatomical and pharmacological studies have been combined to identify the neuronal circuits implicated in the regulation of sleep and wakefulness. We have recently discovered a new investigational technique that has already increased our knowledge of this neural control. Before describing this new technique, however, some basic principles of neuroanatomy may prove useful.
Neuroanatomy: Basic Principles
Modern neuroanatomy is almost entirely at the microscopic level. Even the most powerful microscopes, however, by themselves are insufficient to examine the simplest of neuroanatomical structures. Without proper stains and staining techniques, nuclear cell groups and their connections with other neuroanatomical sites are virtually invisible.
A classic neuroanatomical technique is to inject a "tracer" such as Horseradish Peroxidalse (HRP) into a restricted site in the brain (1). This substance is taken up by the axons that project to the neural structure of interest and is naturally transported by the axon to the cell body. Therefore any cell bodies and axons throughout the brain that are found to contain the tracer HRP are likely to be connected to the neural structure where the injection was made. Now HRP is chemically combined with Wheat Germ Agglutinin (WGA), known as WGA-HRP, to greatly enhance the sensitivity of the methods (2).
This technique would be incomplete without the ability to visualize which axons and cell bodies in the brain contain the tracer. The most common method to do this is known as histochemistry. Typically, the brain is sliced into very thin sections that are washed with a solution that enzymatically colorizes the neural elements containing the tracer. In this way, one can localize cell bodies and axons in successive brain sections and one can map areas of the brain that are connected to a particular neural structure.
We have recently discovered a new tracing method using cholera-toxin instead ot the previously used classical molecules such as HRP and WGA-HRP. In contrast to HRP, which is passively taken up by neurons, cholera-toxin (a bacterial toxin produced by Vibrio cholerae) binds specifically to surface receptors of neurons and is actively taken up and transported by the axons. This phenomenon helps enhance the sensitivity of cholera-toxin as a tracer (3)
Surprisingly, cholera-toxin chemically combined with HRP (CT-HRP) was originally described as a sensitive tracer in the peripheral nervous system in 1977 (4) and 1982 (5) and once had been applied in the central nervous system (6). Cholera-toxin was apparently abandoned as a tracer for two reasons. First, it was found that CT-HRP was only as sensitive as WGA-HRP in the peripheral nervous system. Second, perhaps researchers were reluctant to USC cholera toxin because of its potential toxic properties.
Cholera-toxin is actually composed of two molecular subunits: A and B. Subunit A is responsible for the toxic effect, whereas the non-toxic subunit B is responsible for the internalization and transport of the toxin in axons and cell bodies. Since the B subunit became commercially available, we decided to try it as a tracer. We were the first to inject free cholera-toxin (B subunit) which was not chemically bound with HRP. We were surprised to discover immediately after the first experiment there was a much greater sensitivity with this method relative to WGA-HRP.
The increase in sensitivity that we obtained with this tracer was not only due to its molecular properties, but also due to the method we used to inject it into a neural structure, as well as the staining technique we developed to visualize it. A special injection technique known as iontophoresis, instead of the traditional pressure injection method, permitted us to obtain very restrictcd injection sites of less than 250u in diameter with no necrosis of tissue providing optimal labeling, a performance never before reached by other tracers (7). To visualize the cholera-toxin, we developed an immunohistochemical technique in place of the classical enzymatic "colorizing" histochemical methods used with tracers such as HRP and CT-HRP. With immunohistochemistry, the brain slices are washed with a solution that contains antibodies to cholera-toxin. The antibodies bind specifically to the tracer and~ with proper staining techniques, can be visualized under the microscope.
Summary
The discovery of this new method in 1987 has already lead us to describe previously unknown connections to nuclei classically implicated in the sleep-waking cycle. For example, it was previously thought that the rat locus coeruleus (LC), a nucleus in the brain stem, implicated in sleep wake control, received major projections from only two nuclei (prepositus hypoglossi and paragigantocellularis lateralis). These neural connections were described using WGA-HRP as a tracer. We recently demonstrated using cholera-toxin that the LC also receives major projections from numerous other neural structures (lateral preoptic area and posterior hypothalamic areas, periaqueductal grey and nucleus Kolliker-Fuse). These results highlight the superior sensitivity of cholera-toxin over WGA-HRP as a tracer and indicate that the additional nuclei we described projecting to the LC may be of crucial importance in the control of LC cell discharge and by extension, the sleep-waking cycle. We are currently testing these hypotheses in the lab.
By developing new technologies such cholera-toxin tracer, we may better understand anatomy of sleep controlling centers in the brain. Neuroanatomical mapping has already implicated anatomical structures as potential candidates and may add another piece to the mysterious puzzle of how brain controls sleep and wakefulness.
Pierre Luppi, Ph.D. is a neuroanatomist specializing in neural mechanisms of sleep. Working under the direction of Dr. Jouvet for the past eight years, he is a young upcoming CNRS researcher (equivalent to the NIH in the U.S.A.) He received his doctorate degree neuroscience from the University of Claude Bernard Lyon. His recent discovery of a new neuroanatomical technique has already altered our understanding of the neural control of wakefulness.
|
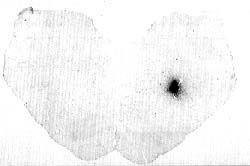
Figure 1 : Photomicrograph illustrating the effects of a microiontophoretic injection of cholera toxin in the pontine reticular formation of the cat. |
References
- La Vail, J.H. and La Vail, M.H.
Retrograde axonal transport in the central nervous system, Science, 176 (1972) 1416. - Gonatas, N.K., Harper, C., Mizutani, T. Gonatas,
J.O.
superior sensitivity of conjugates of horseradish peroxidase with wheat germ agglutinin for studies of retrograde axonal transport, J. Histochem. Cytochem., 27 (1979) 728-734 -
Luppi, P.H., Sakai, K., Salvert, D., Fort, P. and Jouvet M.
Peptidergic hypothalamic afferents to the cat raphe pallidus as revealed by a double immunostaining technique using unconjugated cholera-toxin as retrograde tracer, Brain Research, 402 (1987) 339-345. - Stockel, K., Schwab, M.E., and Thoenen, H.
Role of gangliosides in the uptake and retrograde axonal transport of cholera and tetanus toxin as compared to nerve growth factor and whea agglutinin, Brain Research, 132 (1977) 272-285. - Trojanowski, J.Q., Gonatas, J.O., AND Gonatas
N.K.
Horseradish peroxidase (HRP) conjugates of cholera toxin lectins are more sensitive retrogradely transported markers than free HRP, Brain Research, 231 (1982) 273-285. - Shapiro, R.E. and Miselis, R.R.
The central connections the area postrema of the rat, J. Comp. Neurol., 234 (1985) 344-364 -
Luppi, P-H ., Fort, P. and Jouvet, M.
Iontophoretic application of unconjugated cholera toxin B subunit (CTb) combined with immunohistochemistry of neurochemical substances: a method for transmitter identification of retrogradely labeled neurons. Brain Research, 534 (1990) 209-224.